


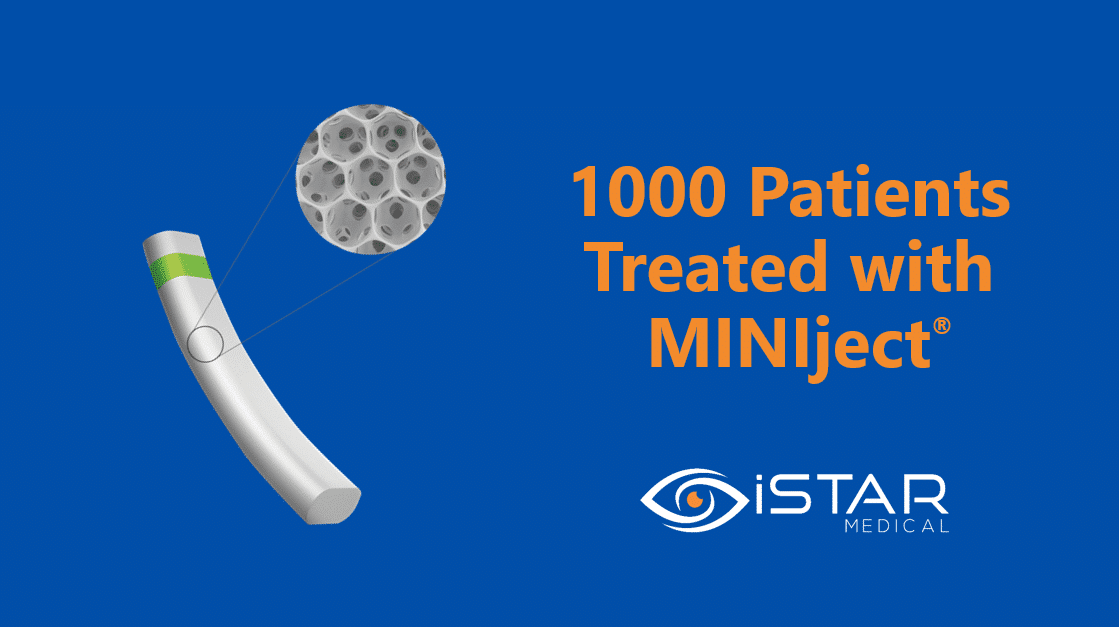
WAVRE, Belgium — 10 May 2023: iSTAR Medical, a medtech company delivering breakthrough eye care solutions to patients, today announces that over 1000 patients have now been treated commercially with MINIject®, its minimally-invasive glaucoma surgery (MIGS) implant. MINIject® is commercially available in Germany, UK, Switzerland, Austria and the Nordic Countries. iSTAR Medical expects further market expansion into additional geographies in 2023.
MIGS procedures close the treatment gap for glaucoma patients between eye drops, which often have side-effects and high rates of non-compliance among patients. The global MIGS devices market is well-established and projected to reach $5 billion by 2030[1]. MINIject® is currently the only commercially available supraciliary MIGS implant, offering a treatment option for mild-to-moderate glaucoma cases having demonstrated meaningful and sustained performance in lowering intraocular pressure (IOP), combined with a favorable safety profile.
said Dr Steven D. Vold, investigator in the STAR-V pre-market approval study to enable commercialization of MINIject® in the U.S.
commented Prof. Dr Kaweh Mansouri, Chief Medical Officer at iSTAR Medical and Consultant Ophthalmologist.
commented Michel Vanbrabant, CEO of iSTAR Medical.
About iSTAR Medical
iSTAR Medical is committed to delivering breakthrough eye care solutions. Our most advanced product, MINIject®, is approved in Europe for the treatment of open-angle glaucoma – the leading cause of irreversible blindness – and we are aiming to seek market approval in the US. We believe MINIject®’s distinctive tissue-integrating capabilities unlock a safer, and more effective option for patients. We are building an exceptional team and pipeline of potentially leading products such as MINIject® to establish new treatment paradigms in eye care conditions with the highest patient needs.
iSTAR Medical is an independent company which entered a strategic partnership with AbbVie (NYSE: ABBV) in July 2022. The collaboration further supports the role of MINIject® in the treatment of glaucoma and accelerates iSTAR Medical’s goal to bring MINIject® to more patients globally while providing AbbVie the opportunity to further expand its diverse eye care portfolio.
About MINIject®
MINIject® is iSTAR Medical’s innovative MIGS device for patients with primary open-angle glaucoma. MINIject® combines the distinctive porous structure of its proprietary STAR material with the power offered by the supraciliary space. As a result, it is designed to enhance natural fluid outflow, reducing intraocular pressure (IOP) and the need for medication, while bio-integrating with surrounding tissue, limiting inflammation, fibrosis and subsequent complications.
About Glaucoma
Glaucoma is a progressive disease affecting over 100 million people globally, of which primary open-angle glaucoma is the most common form.[2,3] IOP reduction, through medication or surgery, helps delay disease progression.[4] Medication is generally the first line treatment, but the progressive addition of multiple drops can burden patients with side effects, compliance challenges and costs.[2,4] Invasive surgery can present risks with irreversible complications and often requires long-term patient management.[2,4] MIGS is the most promising and fastest-growing glaucoma therapy due to its enhanced safety profile.[2] MINIject is potentially best-in-class for its promising long-term efficacy and safety.
[1] Allied Market Research, “Minimally Invasive Glaucoma Surgery (MIGS) Devices Market by Surgery (Glaucoma in Conjunction with Cataract and Standalone Glaucoma), Target (Trabecular Meshwork, Suprachoroidal Space, and Others), Product (MIGS Stents, MIGS Shunts, and Other), and End User (Eye Hospitals, Ophthalmology Clinics, and Outpatient Surgical Centers): Global Opportunity Analysis and Industry Forecast, 2021-2030”, August 2021. https://www.alliedmarketresearch.com/minimally-invasive-slaucoma-surgery-MIGS-devices-market
[2] Market Scope, “2021 Glaucoma Surgical Device Market Report”, July 2021. https://www.market-scope.com/pages/reports/267/2021-glaucoma-surgical-device-market-report
[3] Jonas JB, Aung T, Bourne RR et al. “Glaucoma”. Lancet 2017; 390: 2083–93
[4] “European Glaucoma Society Terminology and Guidelines for Glaucoma”, 4th Edition: British Journal of Ophthalmology. 2017;101:1-195 https://bjo.bmj.com/content/101/5/73
 EN
EN